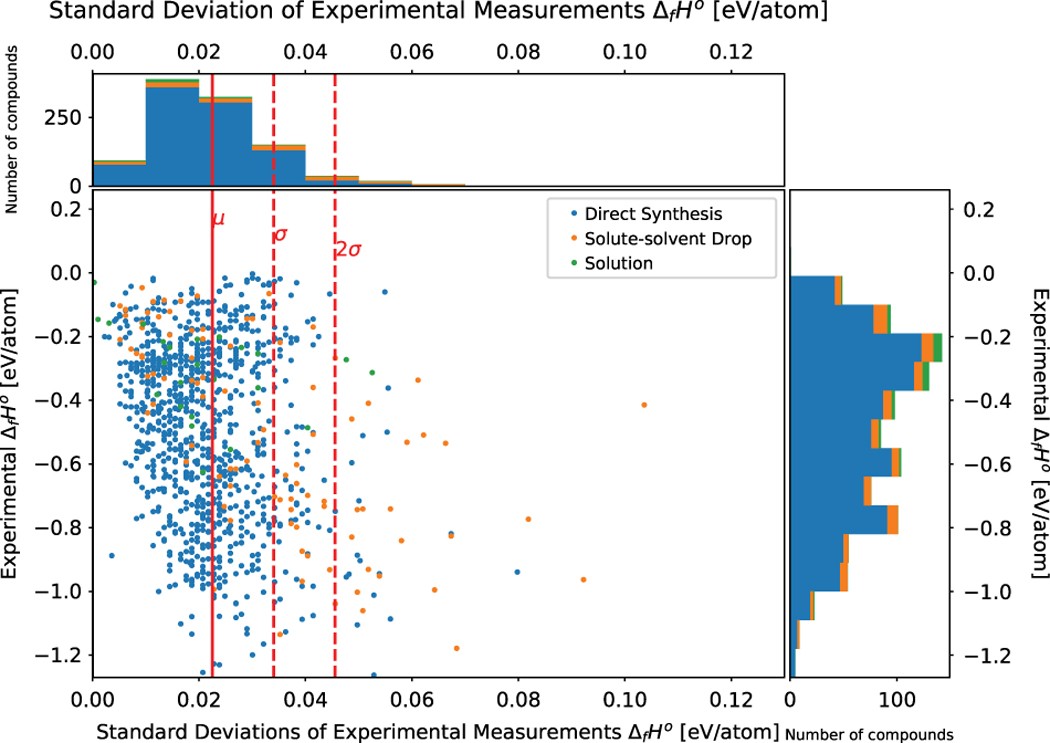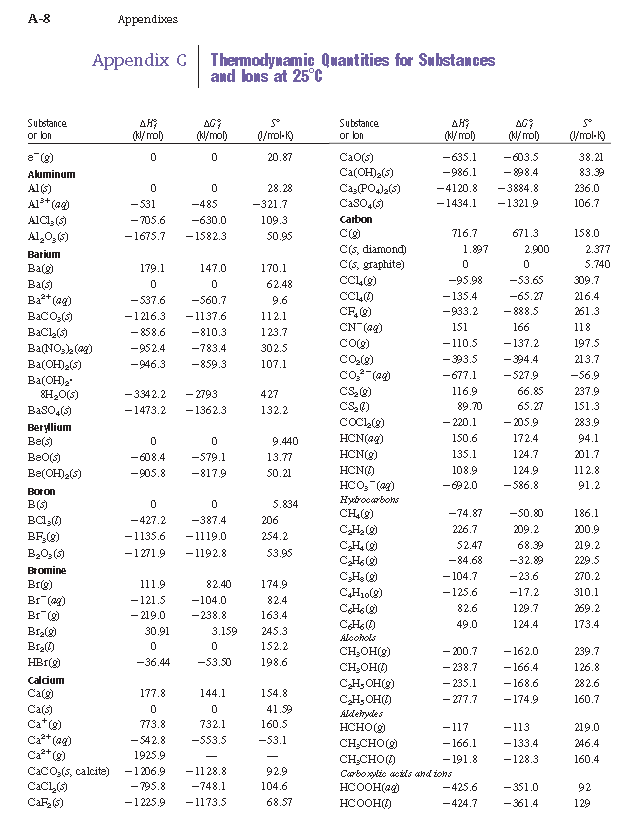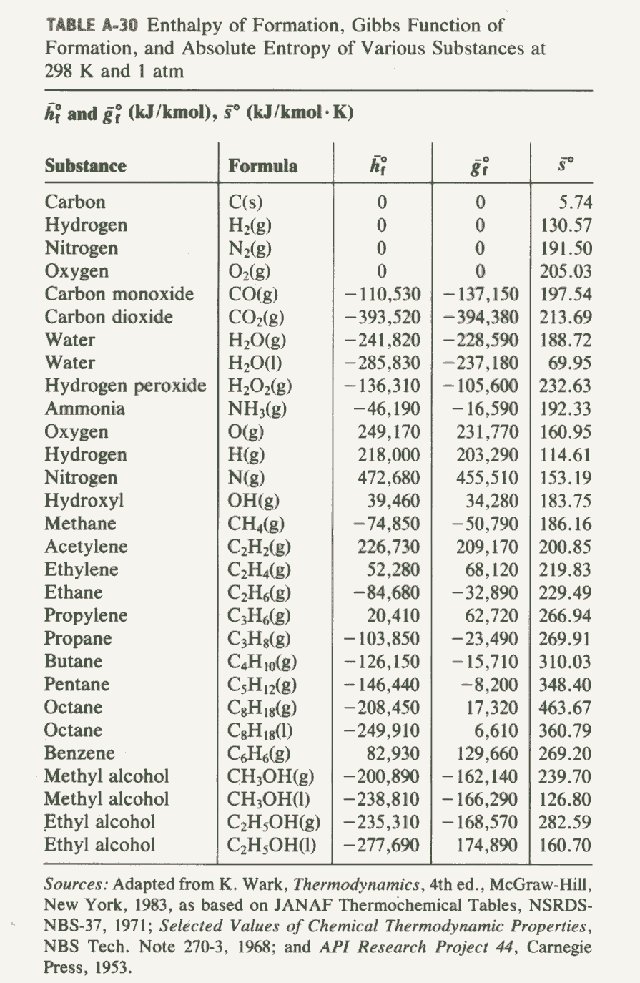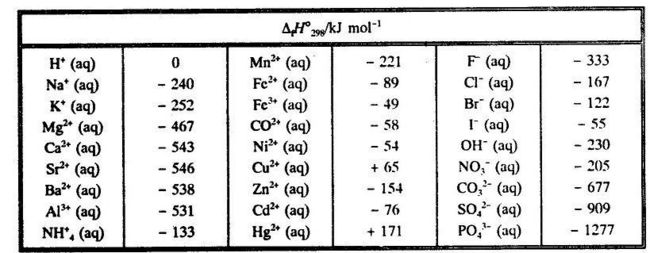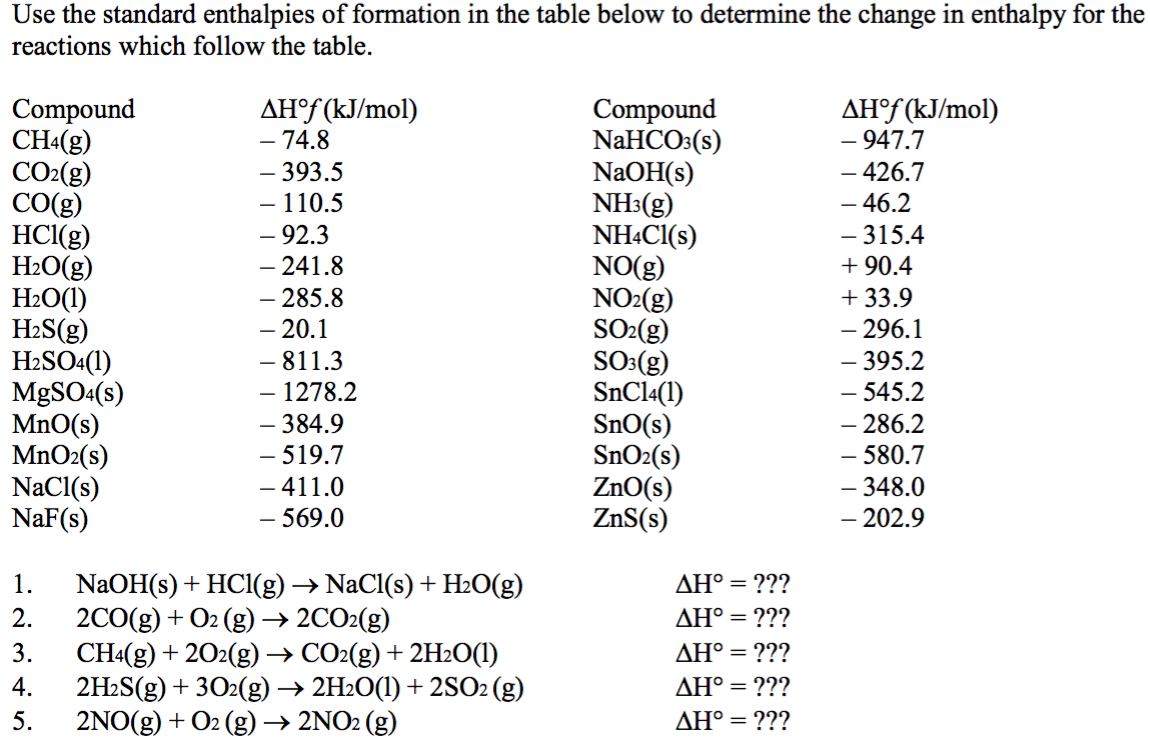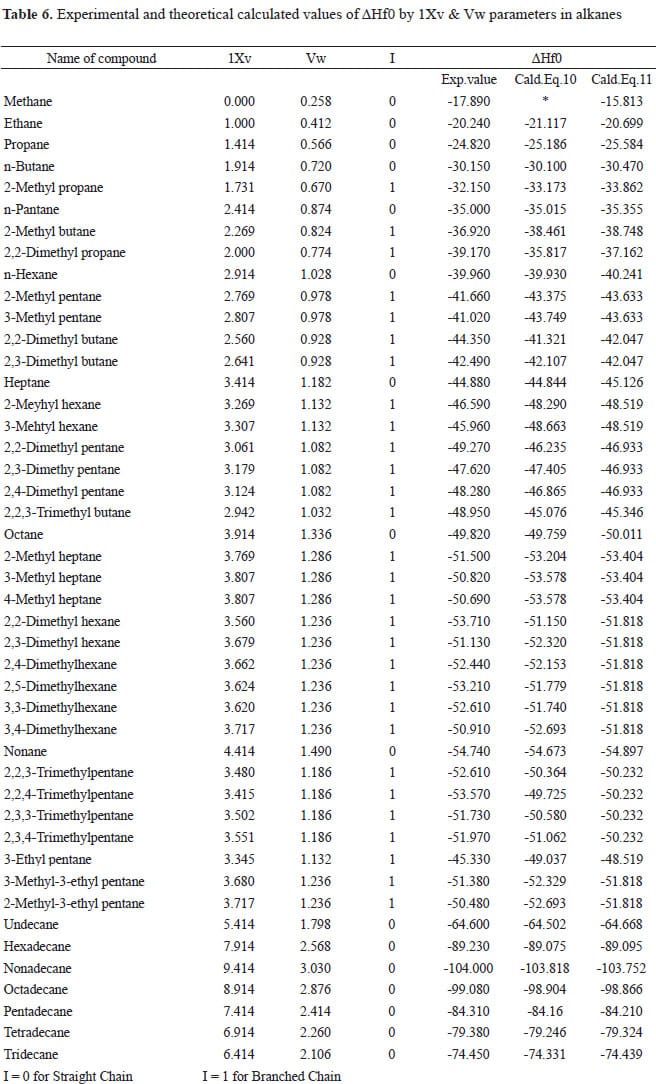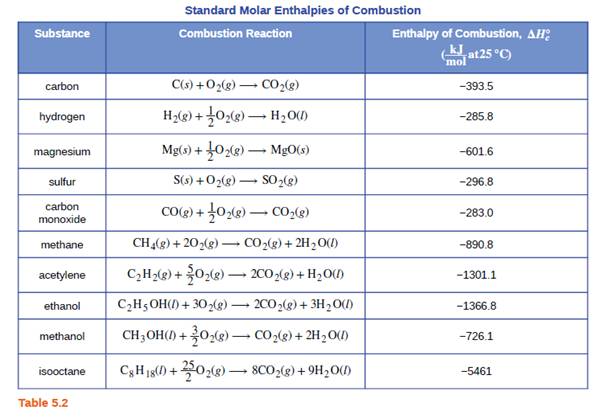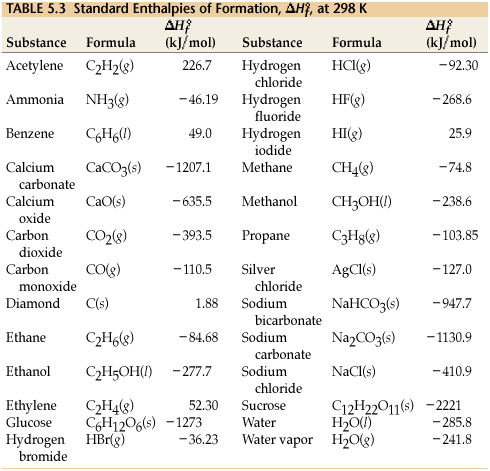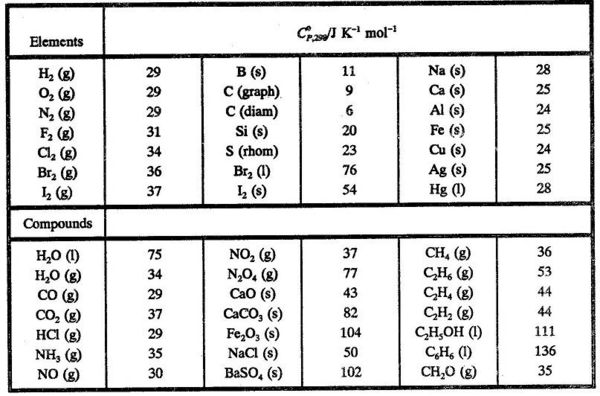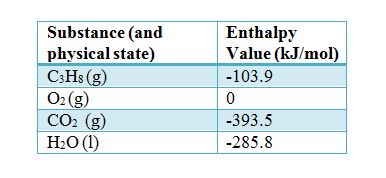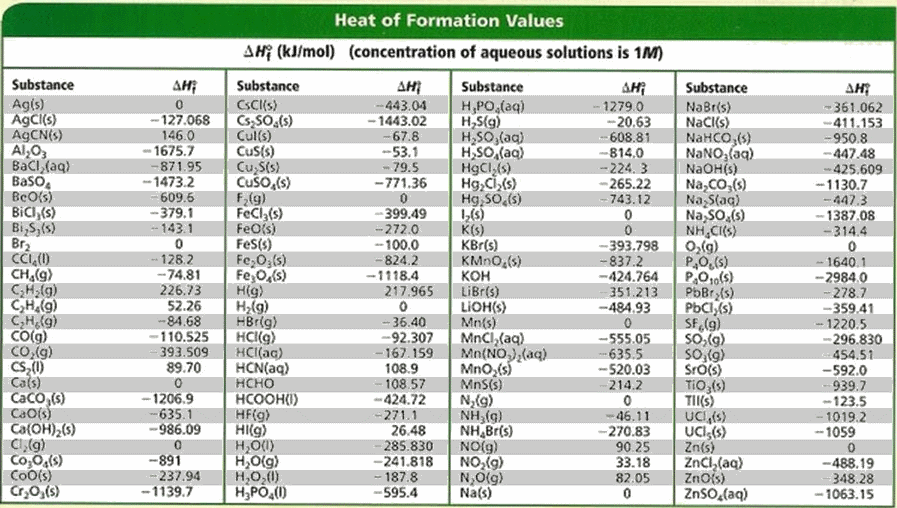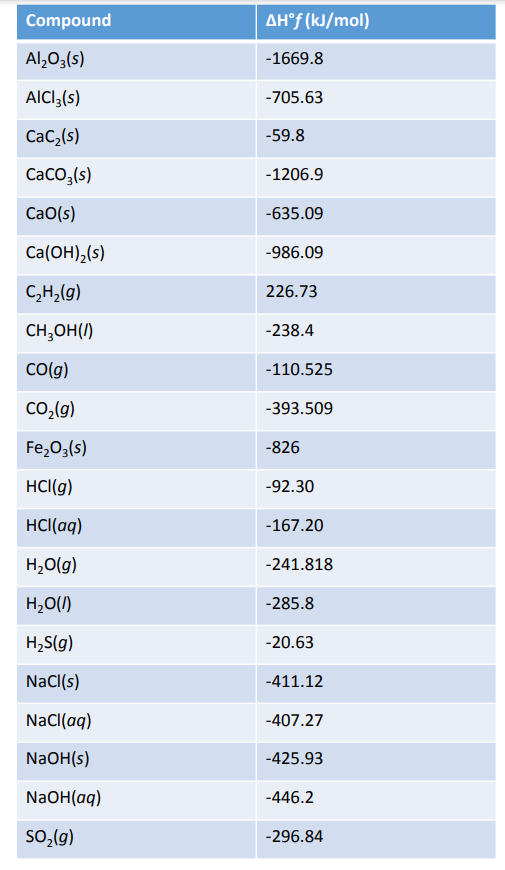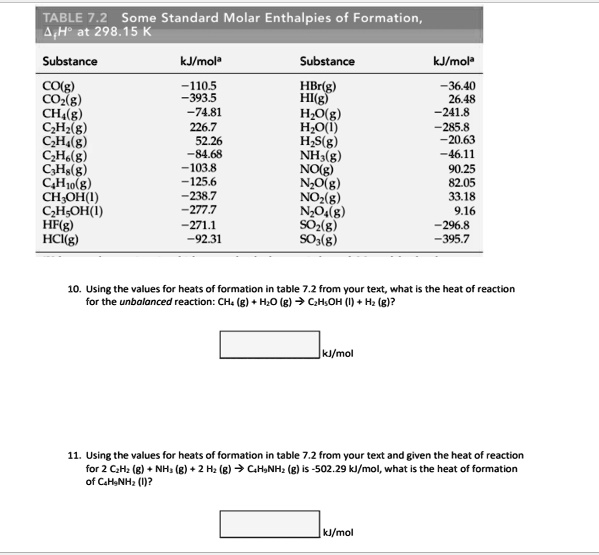
SOLVED: TABLE 7.2 Some Standard Molar Enthalpies of Formation; 0 Heat 298.15 K Substance kJlmol" Substance kJlmol" cO(g= cO-( CHA( CHz CzHA CHa(g- C,Hs(g- C Hj(g) CH,OH() CzHSOH() HF(g) HCIg) -I105 -3935 -
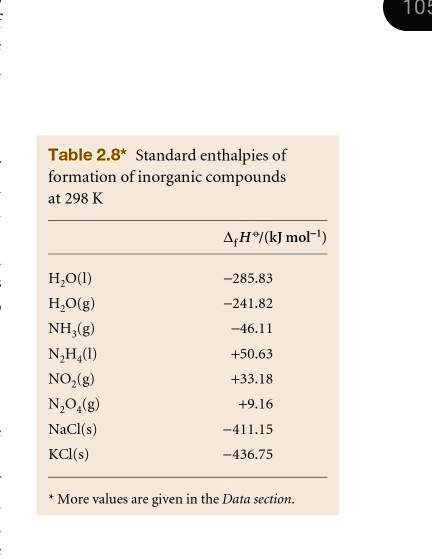
SOLVED: Tu= Table 2.8* Standard enthalpies of formation of inorganic compounds at 298 K 4H"/(kJ mol-!) H,O() HjO(g) NH,(g) N,H() NO(g- N,O,(g) NaCl(s) KCl(s) 285.83 -241.82 -46.14 +50.63 +33.18 19.16 -411.15 -436.75